By
John B. Cornwell and Jeffery D. Marx
Presented At Center for Chemical Process Safety 17th Annual International Conference and Workshop Risk, Reliability, and Security Jacksonville, Florida October 8-11, 2002
Abstract
Although the United States Federal Government does not have a risk-based standard for determining the acceptability of a location relative to the transportation of hazardous materials, a number of state and local regulatory agencies are beginning to require that a risk analysis be performed as part of a siting study. An example of such a legal requirement is the California Code of Regulations, Title 5, Section 14010. This regulation explicitly requires that a risk analysis be performed on hazardous material pipelines and rail cargo transportation routes within 1,500 feet of a proposed public school site.
With only a handful of companies able to perform true Quantitative Risk Analysis (QRA) studies (not qualitative assessments), a method was needed that could apply the results of a series of QRA studies to any proposed location. This can be achieved by developing risk transects for a range of hazardous material pipelines and railcar cargoes. Risk transects create a relationship between individual risk and distance from the pipeline or railway. By varying the commodity, line diameter, and operating pressure for the pipelines, and tank capacity and commodity for the railcars, a set of risk transects can be created that can then be applied to any proposed location. This paper outlines the process of developing the consequence modeling, accident probability data, and risk calculations that are necessary to create risk transects. It also demonstrates how the risk transects can be combined and used to satisfy the risk analysis requirements contained in the California regulations.
Text
Introduction to QRAs for Hazardous Material Transportation

Building a new school in California now requires a risk analysis for the proposed site, subject to California Code of Regulations, Title 5, Division 1, Chapter 13, Subchapter 1, School Facilities Construction, Section 14010. Specifically, Subparts (d) and (h) outline the type of analysis to be performed.
(d) If the proposed site is within 1,500 feet of a railroad track easement, a safety study shall be done by a competent professional trained in assessing cargo manifests, frequency, speed, and schedule of railroad traffic, grade, curves, type and condition of track, need for sound or safety barriers, need for pedestrian and vehicle safeguards at railroad crossings, presence of high pressure gas lines near the tracks that could rupture in the event of a derailment, preparation of an evacuation plan. In addition to the analysis, possible and reasonable mitigation measures must be identified.
(h) The site shall not be located near an aboveground water or fuel storage tank or within 1,500 feet of the easement of an aboveground or underground pipeline that can pose a safety hazard, as determined by a risk analysis study, conducted by a competent professional, which may include certification from a local public utility commission.
Because of the difficult nature of performing a true Quantitative Risk Analysis (QRA) study, a simplified method for applying hazardous material transportation QRA results to any proposed location was needed. By creating generic risk transects for varying pipeline and railcar commodities (with their appropriate properties), the risk at any particular location could be found. Generic risk transects provide a “worst-case” estimate of the risk in the vicinity of a pipeline or railway. Once these are generated, they can be applied to any site as a check for acceptability, since risk transects are a simple relationship between individual risk and distance from the pipeline or railway.
To demonstrate the methodology behind creating this set of risk transects, the hazards for an example set of pipelines and railcars were modeled with the proper consequence modeling, and accident frequencies were developed for potential release with applicable data. These were combined to create a risk transect for each of the following pipelines and railcar systems.
Pipelines
- 30-inch natural gas pipeline operating at 1,000 psig
- 12-inch natural gas pipeline operating at 700 psig
- 8-inch LPG pipeline operating at 450 psig
- 6-inch anhydrous ammonia pipeline operating at 330 psig
- Railcars
- LPG railcar, 33,000 gallons
- Chlorine railcar, 90 tons
- Anhydrous ammonia railcar, 33,000 gallons
Consequence Modeling
From the public’s perception, it is safe to assume that the consequence modeling of potential hazards associated with pipeline and railcar transport of flammable and toxic materials is perceived as more important than the associated statistical analysis of how often a release may occur. After all, if the consequence modeling shows that a release will not impact a proposed school location, then there is no need to continue the risk study. To some degree, the regulations have already limited the range of study by excluding from review any pipeline or railway that is more than 1,500 feet from the proposed school location. However, the effects of many hazardous materials transported by pipeline and railcar, such as liquefied petroleum gas (LPG), anhydrous ammonia, chlorine, etc., can easily reach 1,500 feet or more under a wide range of release and atmospheric conditions. The reasoning behind this limitation in the regulations is not defined and may simply come from “experience.”
Experience will tell the modeling analyst that a release of gas or liquefied gas from any hole size (leak, puncture, or rupture in a pipeline) will result in the formation of a free jet. Jet releases will exhibit rapid mixing of the released material with the surrounding air due to the turbulence generated by the high velocity of the gas (or gas/aerosol mixture) as it enters the surrounding air.
A pressurized release of gas or liquid from a buried pipeline often displaces the overburden (soil), resulting in a free flowing jet of gas entering the atmosphere. Overburden displacement is characterized by the formation of a “crater” around the rupture. Creation of a crater (overburden removal) is dependent on the available “force” that the fluid has upon release. Large releases of medium to high pressure gas or liquid are usually capable of creating a crater. The crater allows the fluid to enter the atmosphere with high velocity and act as a free jet.
A review of several gas pipeline accidents investigated by the United States National Transportation Safety Board (NTSB) indicates that, for gas pipelines with operating pressures above 600 psig, a 1-inch diameter hole will blow away the overburden above the pipeline, resulting in the formation of a crater that allows the gas to escape as an unobstructed free jet. For the purposes of this analysis, the assumption was made that ruptures and punctures (1-inch holes) of the anhydrous ammonia, LPG, and high pressure natural gas pipelines were capable of crater formation, resulting in a free jet release.
A leak or puncture in an anhydrous ammonia, LPG, or chlorine railcar will release the liquefied gas in much the same manner as a release from the pipeline. The primary difference will be that the railcar is above ground; thus, it is not necessary to form a crater and the liquid inventory in the railcar will serve to maintain the flow out of the hole until the liquid level drops below the elevation of the hole.
When performing site-specific consequence analysis studies, the ability to accurately model the release, dilution, and dispersion of gases and aerosols is important if an accurate assessment of potential exposure is to be attained. For this reason, Quest uses a modeling package, CANARY by Quest7, that contains a set of complex models that calculate release conditions, initial dilution of the vapor (dependent upon release characteristics), and the subsequent dispersion of the vapor introduced into the atmosphere. The models contain algorithms that account for thermodynamics, mixture behavior, transient release rates, gas cloud density relative to air, initial velocity of the released gas, and heat transfer effects from the surrounding atmosphere and the substrate. The release and dispersion models contained in the QuestFOCUS package (the predecessor to CANARY by Quest) were reviewed in a United States Environmental Protection Agency (EPA) sponsored study [TRC, 1991] and an American Petroleum Institute (API) study [Hanna, Strimaitis, and Chang, 1991]. In both studies, the QuestFOCUS software was evaluated on technical merit (appropriateness of models for specific applications) and on model predictions for specific releases. One conclusion drawn by both studies was that the dispersion software tended to overpredict the extent of the gas cloud travel, thus resulting in too large a cloud when compared to the test data (i.e., a conservative approach).
A study prepared for the Minerals Management Service [Chang, et al.,1998] reviewed models for use in modeling routine and accidental releases of flammable and toxic gases. CANARY by Quest received the highest possible ranking in the science and credibility areas. In addition, the report recommends CANARY by Quest for use when evaluating toxic and flammable gas releases. The specific models (e.g., SLAB) contained in the CANARY by Quest software package have also been extensively reviewed.
Introduction to Physiological Effects of Fires, Explosions, and Toxic Gas Exposure
The QRAs performed on the pipeline and railway routes involved the evaluation of thousands of potential hazardous material releases. Each potential release may result in one or more of the following hazards.
- Exposure to gas containing a toxic component (e.g., NH3, Cl2).
- Exposure to heat radiation from torch fires (gas released from aboveground pipeline locations, or releases that result from excavation accidents, or releases that result from pipeline ruptures or punctures in which the overburden is blown away and a crater is formed, or a release from a railcar).
- Exposure to explosion overpressure following the release and ignition of liquefied gas (e.g., LPG).
- Exposure to a flash fire following the release and ignition of pipeline gas or liquefied gas.
- Exposure to heat radiation from a Boiling Liquid Expanding Vapor Explosion (BLEVE) of an LPG railcar.
In order to compare the risks associated with each type of hazard listed above, a common measure of consequence must be defined. In risk analysis studies, a common measure for such hazards is their impact on humans. For each of the fire, explosion, and toxic hazards listed, there are data available that define the effect of the hazard on humans.
When comparing a flammable hazard to an overpressure hazard, the magnitude of the hazard’s impact on humans must be identically defined. For instance, it would not be meaningful to compare human exposure to nonlethal overpressures (e.g., low overpressures that break windows) to human exposure to thermal radiation causing second-degree skin burns (e.g., 37.5 kW/m2 for thirty seconds) or to lethal exposure to ammonia vapor (e.g., 3,135 ppm for fifteen minutes)
In this study, risk is defined as the potential exposure of humans to lethal hazards (i.e., radiant heat, overpressure, or toxic gas exposure) that have the potential to occur as a result of accidents originating along the pipeline and railway routes. Lethal exposure levels for the various hazards are discussed in the following sections.
Physiological Effects of Exposure to Thermal Radiation from Fires
The physiological effect of fire on humans depends on the rate at which heat is transferred from the fire to the person, and the time the person is exposed to the fire. Even short-term exposure to high heat flux levels may be fatal. This situation could occur when persons wearing ordinary clothes are inside a flammable vapor cloud (defined by the lower flammable limit) when it is ignited. Persons located outside a flammable cloud when it is ignited will be exposed to much lower heat flux levels. If the person is far enough from the edge of the flammable cloud, the heat flux will be incapable of causing fatal injuries, regardless of exposure time. Persons closer to the cloud, but not within it, will be able to take action to protect themselves (e.g., moving farther away as the flames approach, or seeking shelter inside structures or behind solid objects).
In the event of a continuous torch fire during the release of flammable gas or gas/aerosol, or a pool fire, the thermal radiation levels necessary to cause fatal injuries to the public must be defined as a function of exposure time. This is typically accomplished through the use of probit equations, which are based on experimental dose-response data.
The product ( t∙ Kn) is often referred to as the “dose factor.” According to probit equations, all combinations of intensity (K) and time (t) that result in equal dose factors also result in equal values for the probit (Pr) and therefore produce equal expected fatality rates for the exposed population.
Work sponsored by the U.S. Coast Guard [Tsao and Perry, 1979] developed the following probit relationship between exposure time and incident heat flux.
where:
t = exposure time, sec
I = effective thermal radiation intensity, kW/m2
Table 1 presents the probit results for several exposure times that would be applicable for torch fires and pool fires. The mortality rates and corresponding thermal radiation levels are listed. The graphical form of the thermal radiation probit equation for different exposure times is presented in Figure 1.
|
Exposure Time (sec) |
Probit value |
Mortality Rate* (percent) |
Incident Thermal Radiation Flux | |
|---|---|---|---|---|
| (kW/m2) | (Btu/(hr@ft2)) | |||
| 5 |
2.67 5.00 7.33 |
1 50 99 |
27.87 55.17 109.20 |
8,833 17,485 34,610 |
| 15 |
2.67 5.00 7.33 |
1 50 99 |
12.22 24.20 47.39 |
3,873 7,670 15,178 |
| 30 |
2.67 5.00 7.33 |
1 50 99 |
7.27 14.89 28.47 |
2,304 4,560 9,023 |
| 60 |
2.67 5.00 7.33 |
1 50 99 |
4.32 8.55 16.93 |
1,369 2,709 5,365 |
*Percent of population fatally affected.
Figure 1
Thermal Radiation Probit Relations

The choice of thermal radiation flux levels is influenced by the duration of the fire and the potential time of exposure to the flame by an individual. All combinations of incident heat flux () and exposure time () that result in equal values of “radiant dosage” () produce equal expected mortality rates. An exposure time of 30 seconds was chosen for this analysis for torch fires. People who are exposed to radiant hazards are aware of the hazards and know in which direction to move in a very short period of time.
Physiological Effects of Exposure to Overpressure
The damaging effects of overpressure depend on the peak overpressure that reaches a given structure, and the method of construction of that structure. Similarly, the physiological effects of overpressure depend on the peak overpressure that reaches a person. Exposure to high overpressure levels may be fatal. Persons located outside the flammable cloud when it ignites will be exposed to lower overpressure levels than persons within the flammable cloud. If the person is far enough from the edge of the cloud, the overpressure is incapable of causing fatal injuries.
The vapor cloud overpressure calculations in this analysis were made with the Baker-Strehlow model which is contained in the CANARY by Quest suite of models. This model is based on the premise that the strength of the blast wave generated by a deflagration is dependent on the reactivity of the flammable gas involved; the presence (or absence) of structures, such as walls or ceilings, that partially confine the vapor cloud; and the spatial density of obstructions within the flammable cloud [Baker, et al., 1994, 1998]. This model reflects the results of several international research programs on vapor cloud explosions, which show that the strength of the blast wave generated by a deflagration increases as the degree of confinement and/or obstruction of the cloud increases.
In the event of an ignition and explosion of a flammable gas or gas/aerosol cloud, the overpressure levels necessary to cause injury to the public are often defined as a function of peak overpressure. Unlike potential fire hazards, persons who are exposed to overpressures have no time to react or take shelter; thus, time does not enter into the hazard relationship. Work by the Health and Safety Executive, United Kingdom [HSE, 1991], has produced a probit relationship based on peak overpressure. This probit equation has the following form.
Pr=1.47+1.37ln(p)
where: p = peak overpressure, psig
Table 2 presents the probit results for 1%, 50%, and 99% fatalities. The graphical form of the overpressure probit equation is presented in Figure 2.
| Probit Value |
Mortality Rate* (percent) |
Peak Overpressure | |
|---|---|---|---|
| (psig) | (kPag) | ||
| 2.67 | 1 | 2.4 | 16.55 |
| 5.00 | 50 | 13.1 | 90.73 |
| 7.63 | 99 | 72.0 | 496.83 |
Figure 2
Overpressure Probit Relation

Physiological Effects of Ammonia
Ammonia (NH3) is a colorless, toxic gas with a low threshold limit value (TLV). NH3 is detectable by odor at concentrations much less than those necessary to cause harm. This allows persons who smell the gas to escape. The most serious hazard presented by NH3 is from a large release from which escape is not possible.
A probit equation for NH3 uses the values of -28.33, 2.27, and 1.36 for the constantsandrespectively [Perry and Articola, 1980]. Substituting these values into the general probit equation yields the following probit equation for NH3.
Using the Perry and Articola probit equation given above, the NH3 gas concentration that equates to a 1% fatality rate is 1,131 ppmv for sixty minutes, or 1,883 ppmv for thirty minutes, or 3,135 ppmv for fifteen minutes, etc., as shown in Table 3. Table 3 presents the mortality rates and NH3 concentrations for various exposure times, while Figure 3 presents the same information in graphical form.
Physiological Effects of Chlorine
Chlorine (Cl2) is a greenish-yellow, nonflammable toxic gas with a low threshold limit value (TLV). Cl2 is detectable by a pungent distinctive odor at concentrations much less than those necessary to cause harm. This allows persons who smell the gas to escape. As was the case with ammonia, the most serious hazard presented by Cl2 is from a large release from which escape is not possible.
|
Exposure Time (minutes) |
Probit Value |
Mortality Rate* (percent) |
NH3 Concentration (ppmv) |
|---|---|---|---|
| 5 |
2.67 5.00 7.33 |
1 50 99 |
7,031 14,955 31,809 |
| 15 |
2.67 5.00 7.33 |
1 50 99 |
3,135 6,667 14,182 |
| 30 |
2.67 5.00 7.33 |
1 50 99 |
1,883 4,005 8,519 |
| 60 |
2.67 5.00 7.33 |
1 50 99 |
1,131 2,406 5,117 |
*Percent of exposed population fatally affected.
Figure 3
Ammonia Probit Relations

A probit equation for Cl2 uses the values of -36.45, 3,13, and 2.64 for the constantsandrespectively [Perry and Articola, 1980]. Substituting these values into the general probit equation yields the following probit equation for Cl2.
Using the Perry and Articola probit equation given above, the Cl2 gas concentration that equates to a 1% fatality rate is 24.1 ppmv for sixty minutes, or 31.4 ppmv for thirty minutes, or 40.8 ppmv for fifteen minutes, etc., as shown in Table 4. Table 4 presents the mortality rates and Cl2 concentrations for various exposure times, while Figure 4 presents the same information in graphical form.
|
Exposure Time (minutes) |
Probit Value |
Mortality Rate* (percent) |
Cl2 Concentration (ppmv) |
|---|---|---|---|
| 5 |
2.67 5.00 7.33 |
1 50 99 |
61.8 82.0 108.7 |
| 15 |
2.67 5.00 7.33 |
1 50 99 |
40.8 54.1 71.7 |
| 30 |
2.67 5.00 7.33 |
1 50 99 |
31.4 41.6 55.1 |
| 60 |
2.67 5.00 7.33 |
1 50 99 |
24.1 32.0 42.4 |
*Percent of exposed population fatally affected.
Accident Frequencies
The likelihood of a particular accident occurring within some specific time period can be expressed in different ways. One way is to state the statistical probability that the accident will occur during a one-year period. This annual probability of occurrence can be derived from failure frequency data bases of similar accidents that have occurred with similar systems or components in the past.
Most data bases (e.g., CCPS [1989], OREDA [1984]) that are used in this type of analysis contain failure frequency data (e.g., on the average, there has been one failure of this type of equipment for 347,000 hours of service). By using the following equation, the annual probability of occurrence of an event can be calculated if the frequency of occurrence of the event is known.
Figure 4
Chlorine Probit Relations

Chlorine Probit Relations
If an event has occurred once in 347,000 hours of use, its annual failure frequency is computed as follows.
The annual probability of occurrence of the event is then calculated as follows.
Note that the frequency of occurrence and the probability of occurrence are nearly identical. (This is always true when the frequency is low.) An annual probability of occurrence of 0.0249 is approximately the same as saying there will probably be one event per forty years of use.
Due to the scarcity of accident frequency data bases, it is not always possible to derive an exact probability of occurrence for a particular accident. Also, variations from one system to another (e.g., differences in design, operation, maintenance, or mitigation measures) can alter the probability of occurrence for a specific system. Therefore, variations in accident probabilities are usually not significant unless the variation approaches one order of magnitude (i.e., the two values differ by a factor of ten).
The following subsections describe the basis and origin of failure frequency rates used in this analysis.
Pipeline Failure Rate Data
Department of Transportation (DOT) data for underground liquid pipelines in the United States indicate a failure rate of 1.35 x 10‑3 failures/mile/year [DOT, 1988]. Data compiled from DOT statistics on failures of gas pipelines show a failure rate of 1.21 x 10‑3 failures/mile/year for steel pipelines in the United States [Jones, et al., 1986]. In addition to failures of buried pipe, these data include failures of buried pipeline components, such as block valves and check valves, when the failure resulted in a release of fluid from the pipeline.
These data sets are not sufficiently detailed to allow a determination of the failure frequency as a function of the size of the release (i.e., the size of hole in the pipeline). However, British Gas has gathered such data on their gas pipelines [Fearnehough, 1985]. These data indicate that well over 90% of all failures are less than a one-inch diameter hole, and only 3% are greater than a three‑inch diameter hole.
Data compiled from DOT data on gas pipelines in the United States show a trend toward higher failure rates as pipe diameter decreases [Jones, et al., 1986]. (Smaller diameter pipes have thinner walls; thus, they are more prone to failure by corrosion and by mechanical damage from outside forces.)
Railcar Failure Rate Data
Railroad accidents can result from a number of causes; track defects, operational errors, train control equipment failure (e.g., signaling), etc. For the purposes of the risk analysis, the specific cause of an accident that results in a release of product is not specifically required. The use of historic data to define the accident failure rate will be sufficient.
The construction of the QRA will assume a Track Class of 3 (30 mph maximum speed) for the analysis. Track classes range from 1 (10 mph) to 6 (110 mph) according to the Federal Railroad Administration Safety Standards (49 CFR 213). Class 3 track was assumed to represent track that would be located near most schools in urban or suburban areas.
The historical data yield a derailment rate of approximately 5.0 x 10-7 derailments per loaded mile for Class 3 track [FRA, 1992]. When this derailment rate is combined with the conditional probability that a release occurs from a pressurized rail car (e.g., LPG, Cl2, or NH3) at the rate of 0.08 releases/derailment, the release probability becomes 4.0 x 10-8 releases from a pressurized railcar per loaded mile traveled.
The chance of a BLEVE occurring following a derailment and release of flammable material is determined by a number of factors: size and duration of the release, ignition of the released material, orientation of the ignited jet, and impingment of the flame on the railcar shell. Although we are not aware of specific historical data that would provide a probability of BLEVE per loaded mile traveled, for the purposes of this example, it was assumed 5% of the flammable pressurized railcar derailments that caused a release of cargo eventually BLEVEd. This results in a railcar BLEVE probability of 2.0 x 10-9 BLEVEs per mile for a loaded LPG railcar.
Risk Quantification
Conceptually, performing a risk analysis for each pipeline or railway route near a school site is straightforward. For example, for releases that involve toxic or flammable materials, the analysis can be divided into the following steps.
- Along each section of the pipeline or railway, determine the potential credible releases that woold create a toxic or flammable gas cloud, vapor oud explosion, torch fire, pool fire, or BLEVE.
- Determine the probability of occurrence of each of these releases.
- Calcolate the size of each potentially fatal hazard zone created by each of the releases identified in Step 1.
- The hazards of interest are:
- Toxic vapor clouds
- Fire radiation from flash fires, torch fires, pool fires, and BLEVE fireballs
- Overpressures from vapor cloud explosions
- The size of each hazard zone is a function of one or more of the following factors.
- Orientation of the release
- Wind speed
- Atmospheric stability
- Local terrain
- Composition, pressure, and temperature of the fluid being released
- Hole size
- Pipeline or railcar inventory
- Diameter of flammable/combustible liquid pool (if any)
- The hazards of interest are:
- Determine the risk in the vicinity of the pipeline.
- For a specific wind direction, the potential exposure of any individual to a specific hazard zone depends on the following factors.
- Size (area) of the hazard zone
- Location of the individual (i.e., on the school site) relative to the release location
- Wind direction
- Determine the exposure of areas along the pipeline or railway route to each potential hazard zone.
- Perform toxic vapor cloud, flash fire, torch fire, and vapor cloud explosion hazard zone calcolations for all wind directions, wind speeds, atmospheric stabilities, terrain conditions, and release orientations.
- Perform torch fire and pool fire hazard zone calcolations for all wind speeds and wind directions.
- P(wd,ws,stab) ‘ probability that the wind blows from a specified direction (wd), with a certain wind speed (ws), and a given atmospheric stability class, A through F (stab). Meteorological data are generally divided into sixteen wind directions, six wind speed classes, and six Pasquill-Gifford atmospheric stability categories. Although all 576 combinations of these conditions do not exist, a significant number will exist for each release studied. Figure 5 represents the wind speed versus stability distribution for the meteorological data.
- P(acc) ‘ probability of occurrence of each release identified in Step 1.
- P(ii) ‘ probability of immediate ignition (i.e., probability that ignition occurs nearly simoltaneously with the release).
- P(di) ‘ probability of delayed ignition (i.e., probability that ignition occurs after a vapor cloud has formed).
- P(vce) ‘ probability that the vapor cloud explodes following delayed ignition.
- P(orientation) ‘ probability that hazardous fluid is released into the atmosphere in a particolar orientation.
- Sum the potential exposures from each of the hazards for all releases identified in Step 1. This summation requires modifying each potential hazard zone by its probability of occurrence (i.e., the probability of a specific vapor cloud explosion is P(acc)@P(orientation)@ P(ws,wd,stab)@P(di)@P(vce)).
Modify each of the above exposures by its probability of occurrence. Probabilities are divided into the following groups.
Figure 5
Example Wind Speed/Atmospheric Stability Categories

Risk Analysis Results
Individual Risk Transects
Individual risk transects (risk as function of distance from the pipeline or railway) were constructed for the four example pipelines and three example railcar cargoes. Examples from the following scenarios are presented in this section of the report.
- release from a 30-inch, 1,000 psig, natural gas transmission pipeline
- release from a 12-inch, 700 psig, natural gas distribution pipeline
- release from an 8-inch, 450 psig, LPG transmission pipeline
- release from a 6-inch, 330 psig, anhydrous ammonia transmission pipeline
- derailment and release from an LPG railcar, 33,000-gallon capacity
- derailment and release from an anhydrous ammonia railcar, 33,000-gallon capacity
- derailment and release from a chlorine railcar, 90-ton capacity
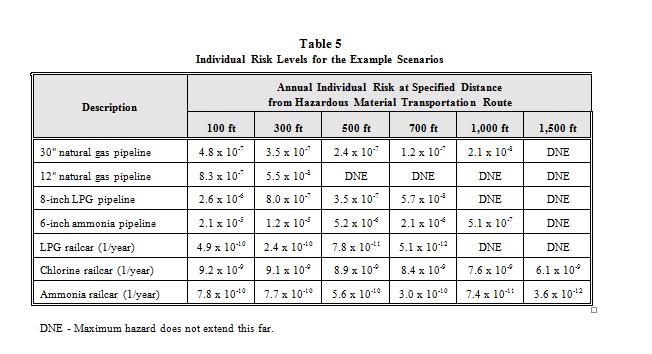
Individual risk calculations present levels of risk based on annual exposure. For any risk level identified at a specific location, that level of risk is based (conservatively) upon one’s presence 24 hours a day, 365 days per year. The meaning of a particular value of risk at a location is related to the hazards that are modeled and their probability. For example, if an individual in the vicinity of a pipeline is at a location that corresponds to a 1.0 x 10-6 risk level, then his risk from being fatally affected by any one of the potential releases from that pipeline is one chance in one million per year. At a location closer to the pipeline, the risk is higher, and farther away from the pipeline, the risk is lower. Table 5 presents the individual risk levels found at specific distances from the seven example pipeline and railcar scenarios. For the railcar scenarios, the calculated risk is based on one (1) loaded railcar passing by the location per year. To adjust for the true number of railcars passing by a site, the number of railcars per year is simply multiplied by the individual risk level.
There are several site-specific factors that can affect the final risk values that are calculated. In most cases, the site-specific factors will only affect portions of the analysis and thus will not be able to influence the
Table 5
Individual Risk Levels for the Example Scenarios
analysis to a large degree. One example is the use of site-specific weather data. Using southern California as an example, the weather patterns in Los Angeles are not significantly different than those in Ventura. If two sets of risk transects are developed, one using Los Angeles weather data and one using Ventura weather data, very little difference would be found between the two. The reason for this is in the frequency data. Small differences in weather data (speed, direction, and stability) do not significantly affect the risk calculations. This is because the risk calculations take into account all the weather combinations (and their associated frequencies), not just the “worst case.” Thus, decreasing the frequency of one particular weather triplet (speed, direction, and stability) increases the frequency of a different weather triplet. In the end, the risk sum may be slightly different, but rarely to the extent that is significant.
Site-specific factors should be evaluated on a case-by-case basis. One example would be some type of unusual terrain feature, such as a tall hill between the proposed school site and a pipeline corridor. If the pipeline corridor only transports natural gas, then it may be possible to make the argument that the effects of a pipeline release do not impact the school.
Individual Risk Transects-Pipeline Sections
Plots of individual risk were constructed for the four example pipelines. Each pipeline evaluated has a different individual risk transect due to differences in physical dimensions (e.g., line size), operating conditions (e.g., line pressure), fluid compositions, and failure frequencies. For each pipeline, the risk of being exposed to a fatal hazard due to a release of fluid along the pipeline is presented as a function of distance from the pipeline.
For the four example pipelines, the individual risk values are presented in tabular form in Table 5 and graphical form in Figure 6.
Figure 6
Risk Transects for the Example Pipelines

The following observations can be made from Figure 6.
(a) If an individual were continuously standing on top of the 12-inch natural gas pipeline for one year, he would have a slightly greater than 1.0 x 10‑6 (one in one million) chance of being killed by a release from the pipeline during that year. The release could be from a corrosion hole, puncture, or rupture of the line that resulted in a continuous gas fire, flash fire, or vapor cloud explosion. A similar result can be drawn from the three other pipeline risk transects.
(b) As distance from the pipeline increases, the risk of fatality due to a release from any one of the pipelines decreases.
(c) At distances greater than about 400 feet from the 12-inch natural gas pipeline, there are no lethal hazard impacts; thus, the risk of fatality due to releases from the pipeline falls to zero. The same is true for the LPG pipeline at distances greater than about 1,000 feet; greater than about 1,200 feet for the 30-inch natural gas line; and greater than 1,400 feet for the anhydrous ammonia pipeline.
Individual Risk Transects – Railway Lines
The base cases for the individual risk transects for the railway lines are calculated assuming one loaded railcar passes by the school site per year. Risk transects for the three example rail cargoes are presented in Figure 7. A review of Figure 7 reveals the following.
(a) The risk of being fatally affected by a release from a chlorine railcar (one passing railcar per year) is approximately 9.0 x 10-9 near the rail line and decreases very little out to 1,500 feet from the rail line. This is due to the long distances traveled by chlorine gas in a potentially lethal concentration. Large releases can extend much farther than the 1,500 foot distance set by the regulations.
Figure 7
Risk Transects for the Three Example Rail Cargoes (1 railcar/year)
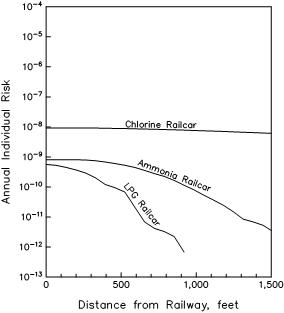
(b) Peak risk levels from one loaded ammonia or LPG railcar per year are approximately 8.0 x 10-10 near the railway and decrease significantly as distance from the railway increases.
(c) At distances greater than about 1,000 feet from the rail line, there are no lethal LPG railcar hazard impacts (hazards are non-lethal at this distance), and thus no risk of fatality due to releases from an LPG railcar. Hazards for ammonia and chlorine railcar releases extend well past the 1,500 foot cutoff distance.
If we compare the risk transects for the LPG pipeline and LPG railcar, we see that the shapes of the curves are similar, which shows that the hazards from a pipeline release and from a damaged railcar are similar. But, there is a considerable difference in the frequency of these events. If we compare the peak risk (the risk on top of the pipeline or on the rail line), the pipeline is about 6,500 times as likely to create a fatal hazard than the one railcar that passes by each year. This means that it would take about 6,500 railcars per year passing by the school site to create the same risk as the pipeline. The total LPG railcar risk is a simple multiplication of the number of railcars and the previously calculated risk for one railcar per year. So once the true number of LPG railcars passing by the school site in one year is known, the risk transect can be adjusted to reflect the true total risk.
Application of Risk Transects to School Siting
The development of a range of pipeline and railcar risk transects provides a consistent approach to evaluating the potential risk to a proposed school location. Since the pipeline and railcar risk transects are independent, their individual contributions are additive when evaluating the total risk to a proposed school location. This is best demonstrated by a series of examples.
- Proposed School Site is Located 330 Feet from a 30-inch Natural Gas Transmission Pipeline
A review of Table 5 and Figure 6 would indicate that the individual risk at the school site is approximately 3.3 x 10-7/year.
Using an individual risk acceptability standard of 1.0 x 10-6 fatality/year (one chance in one million per year of being fatally affected by a release from the pipeline) would indicate that the school site is within (below) the acceptable range.
- Proposed School Site is Located 330 Feet from a 30-inch Natural Gas Transmission Pipeline and an 8-inch LPG Pipeline Laid in the Same Corridor
Using the data presented in Table 5 and the risk transects for the individual pipelines (Figure 6), it can be seen that neither pipeline individually produces a risk level above 1.0 x 10-6 per year at the school site. However, when contributions from the two pipelines are summed, the risk level at the school site becomes 1.03 x 10-6. If the acceptable limit is 1.0 x 10-6, this site would not be deemed acceptable. A more detailed analysis to generate a better estimate of the true risk may be required for this site.
- Example 3. Proposed School Site is Located 850 Feet from a Railway
A railway runs near the boundary of the proposed school site. Rail officials have provided the following information about the recent rail activity along the railway section by the school site for the three example cargoes.
- 1,335 loaded LPG railcars per year
- 240 loaded NH3 railcars per year
- 43 loaded Cl2 railcars per year
Using this activity data, the risk transects for the three example cargoes can be constructed. These are presented in Figure 8, along with a summation risk transect. The summation transect represents the risk from all three cargoes at any distance from the rail line. As can be seen from the figure, the proposed school site at 850 feet from the railway would be acceptable using the 1.0 x 10-6 criteria. If the school were within 250 feet of the railway, the combined risk from the three rail-transported commodities would not be acceptable.
- Proposed school Site is Located Near a Pipeline Corridor and a Railway
In some cases, such as the example in Figure 9, a school site may be proposed in a location that can be affected by both pipeline and railcar releases. In this event, the impacts of one transportation mode on the other are assumed to be independent and the potential risks are additive. In the example calculation, the pipeline corridor (with a 30-inch natural gas distribution line and a 6-inch anhydrous ammonia line) is located 900 feet from the edge of the school property. On the other side of the school site is a railway (1,040 feet from the nearest school boundary), with the following annual activity.
- 522 loaded LPG railcars per year
- 95 loaded NH3 railcars per year
- 36 loaded Cl2 railcars per year
Figure 8
Risk Transects for the Railway in Example 3

The risk data for the school site are presented in Table 6. The summation of risk from all five sources is calculated for the northeastern most point of the school property. This is the location on the proposed site that will experience the highest individual risk level. All other locations at the site will be at a greater distance from the pipeline corridor, the rail line, or both, so the individual risk will be lower. Based on the 1.0 x 10-6 acceptability criteria, this proposed site would not be deemed acceptable.
Summary
A method has been developed that provides a consistent and defensible approach for evaluating the potential impact of hazardous material pipelines and railcar cargoes on potential school sites. The methodology satisfies the State of California’s Code of Regulations for evaluating potential schools sites. In addition, because of the modular nature of the methodology, additional pipeline systems and railcar cargoes can be added to the system. If it were to become necessary, road transportation routes and cargoes could easily be added to the system since the procedure for developing roadway risk transects would be similar to that used for the railway risk transects.
The results of risk transect calculations (risk versus distance) can easily be incorporated into a simple spreadsheet program where site-specific modifiers, such as distance from the pipeline or railway to the school site, number of railcar cargoes, etc., would be input parameters. Such an approach would allow the system to be used throughout the school system network and avoid the widely varying results currently generated by qualitative analyses.
Figure 9
Area Surrounding the Proposed School Site for Example 4
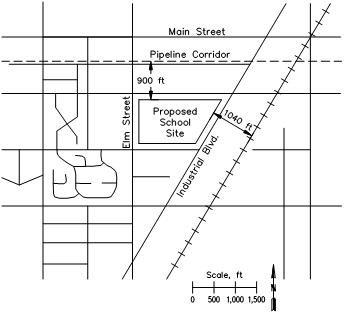
Table 6
Individual Risk Values for Example 4

References
- Baker, Q. A., M. J. Tang, E. Scheier, and G. J. Silva (1994), “Vapor Cloud Explosion Analysis.” Presented at the 28th Loss Prevention Symposium, American Institute of Chemical Engineers (AIChE), April 17-21, 1994.
- Baker, Q. A., C. M. Doolittle, G. A. Fitzgerald, and M. J. Tang (1998), “Recent Developments in the Baker-Strehlow VCE Analysis Methodology.” Process Safety Progress, 1998: p. 297.
- CCPS (1989), Guidelines for Process Equipment Reliability Data, with Data Tables. Center for Chemical Process Safety of the American Institute of Chemical Engineers, 345 East 47th Street, New York, New York, 1989.
- Chang, Joseph C., Mark E. Fernau, Joseph S. Scire, and David G. Strimaitis (1998), A Critical Review of Four Types of Air Quality Models Pertinent to MMS Regulatory and Environmental Assessment Missions. Mineral Management Service, Gulf of Mexico OCS Region, U.S. Department of the Interior, New Orleans, November, 1998.
- DOT (undated), “Computer Printout of Accident Reports, 1971 through 1979.” Submitted to the Office of Pipeline Safety, Department of Transportation, DOT Form 7000-1.
- ERCB (1990), Risk Approach: An Approach for Estimating Risk to Public Safety from Uncontrolled Sour Gas Releases (Volume 6). Energy Resources Conservation Board, Report 90-B, Calgary, Alberta, Canada, October, 1990.
- Fearnehough, G. D. (1985), The Control of Risk in Gas Transmission Pipelines. British Gas Research and Development, D.473, January, 1985.
- FRA (1992), Railroad Accident/Incident Database, 1992. U.S. Federal Railroad Administration, Office of Safety, 400 7th Street S.W., Washington, D.C., 20590, 1992.
- Hanna, S. R., D. G. Strimaitis, and J. C. Chang (1991), Hazard Response Modeling Uncertainty (A Quantitative Method), Volume II, Evaluation of Commonly-Used Hazardous Gas Dispersion Models. Study cosponsored by the Air Force Engineering and Services Center, Tyndall Air Force Base, Florida, and the American Petroleum Institute; performed by Sigma Research Corporation, Westford, Massachusetts, September, 1991.
- HSE (1991), Major Hazard Aspects of the Transport of Dangerous Substances. Health and Safety Executive, Advisory Committee on Dangerous Substances, London, United Kingdom, 1991
- Jones, D. J., G. S. Kramer, D. N. Gideon, and R. J. Eiber (1986), An Analysis of Reportable Incidents for Natural Gas Transmission and Gathering Lines, 1970 through June 1984. Prepared for the Pipeline Research Committee, American Gas Association, NG-18, Report No. 158, March, 1986.
- OREDA (1984), OREDA, Offshore Reliability Data Handbook (First Edition). OREDA, Post Office Box 370, N-1322 Hovik, Norway, 1984.
- Perry, W. W., and W. P. Articola (1980), “Study to Modify the Vulnerability Model of the Risk Management System.” U.S. Coast Guard, Report CG-D-22-80, February, 1980.
- TRC (1991), Evaluation of Dense Gas Simulation Models. Prepared for the U.S. Environmental Protection Agency by TRC Environmental Consultants, Inc., East Hartford, Connecticut 06108, EPA Contract No. 68-02-4399, May, 1991.
- Tsao, C. K., and W. W. Perry (1979), Modifications to the Vulnerability Model: A Simulation System for Assessing Damage Resulting from Marine Spills. U.S. Coast Guard Report CG-D-38-79, Washington, D.C., March, 1979.






